Liver Health Made Simple,
Safe, and Non-Invasive
Visualizing the Invisible with LIVERFASt™
What We Test:
Fibrosis
Activity
Steatosis

What You Get:

Screening

Staging

Monitoring
Our solutions are medical devices, developed with the help of artificial intelligence and advanced analytics. They have been validated by leading clinicians over many years and are easy to do.
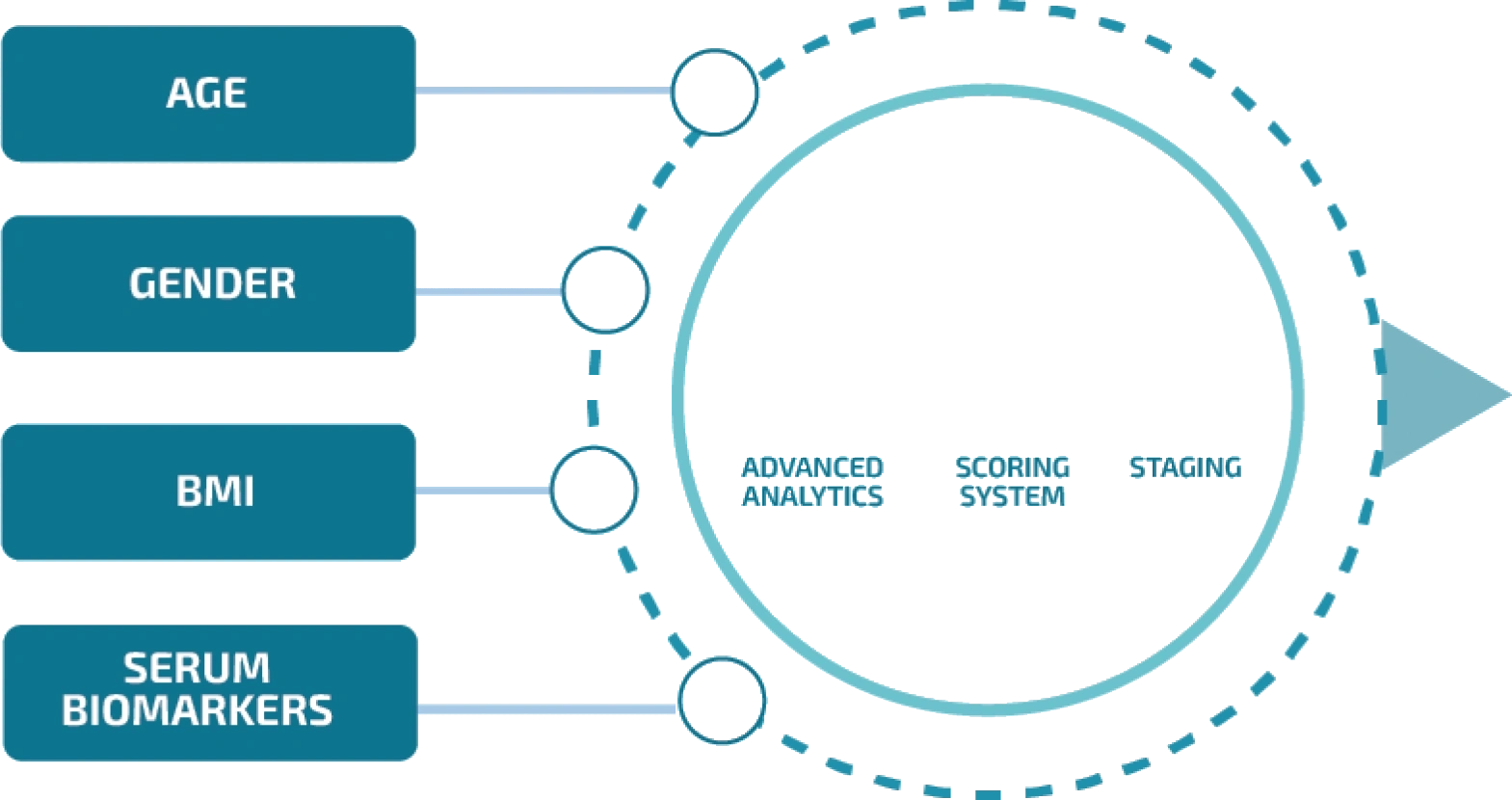
What is LIVERFASt™?
LIVERFASt™ is a blood-based noninvasive test. It uses the input from 10 biomarkers and patient anthropometrics to generate and provide results using advanced analytics and a scoring system that has been developed over 10 years of research.
What does LIVERFASt™ Measure?
LIVERFASt™ uses the input from 10 biomarkers and patient anthropometrics to generate a score from 0.00 to 1.00. The score corresponds to the fibrosis, activity and steatosis staging, similar to a liver biopsy. In doing so, LIVERFASt™ provides high quality diagnostics information without the need for invasive procedures like a biopsy.
Fibrosis

STAGE
INTERPRETATION
FO
No fibrosis
F1
Mild fibrosis
F2
Advanced fibrosis
F1
Significant fibrosis
F4
Severe fibrosis/cirrhosis
Activity

STAGE
INTERPRETATION
aO
No activity
a1
Mild activity
a2
Moderate activity
a1
Marked activity
a4
Severe activity
Steatosis

STAGE
INTERPRETATION
sO
No steatosis (<5%)
s1
Mild steatosis(5-33%)
s2
Moderate steatosis(34-66%)
s3
Marked steatosis (≥ 67%)
LIVERFASt™ in Numbers
Over
Sold in
Adopted by
Did you know?
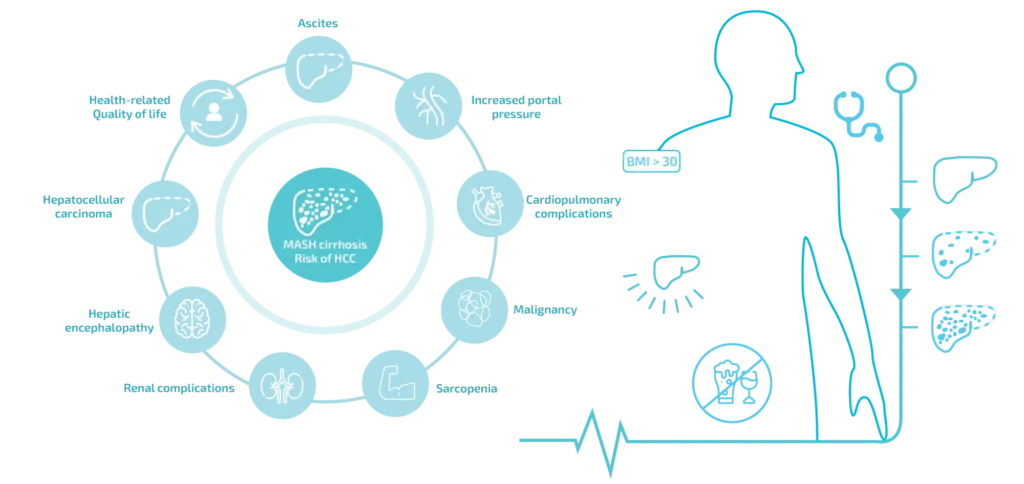
MASH & MASLD (formerly known as NASH & NAFLD) are metabolic diseases of liver. MASH & MASLD can affect people of any age, including children. It is more common in people who live with certain conditions, including obesity (BMI >30) and conditions that may be related to obesity, such as type 2 diabetes, high blood pressure or cholesterol.
How it Works
LIVERFASt™ is easy to do. A simple blood draw from a patient is analyzed in Fibronostics’ CAP & CLIA certified laboratory. Results are returned to the prescribing physician via our portal, physician’s EMR or other forms of integration.

Physician prescribes
LIVERFASt™

Lab analyzes
10 biomarkers from
1 blood draw

Biomarker values plus age,
height, weight, gender, are
entered into the platform

LIVERFASt™ results
are available

Physician prescribes
LIVERFASt™

Lab analyzes
10 biomarkers from
1 blood draw

Biomarker values plus age,
height, weight, gender, are
entered into the platform

LIVERFASt™ results
are available
immediately
Our Benefits
Fast and
Easy-to-do
Minimized
waiting time
Noninvasive
Only one blood draw
required to run our test
Accurate
Robust testing and
validation
Suitable for Multiple Care Settings
Suitable for both early to
late stage care
Benefits

Fast and
Easy-to-do
Minimized
waiting time
Noninvasive
Only one blood draw
required to run our test

Accurate
Robust testing and
validation

Suitable for Multiple Care Settings
Suitable for both early to
late stage care
Your Trusted Partner in Liver Health
Discover LIVERFASt in an Informational Video
Interested in LIVERFASt™?
Contact our sales representative to inquire more about LIVERFASt™ and how it can help you and your patients improve their liver health.